July 29, 2018
It doesn’t often happen that army generals switch sides in the middle of a war, but when cancer’s attack is underway, it may even cause a gene that acts as the body’s master defender to change allegiance.
New research from the Weizmann Institute of Science and reported recently in the Proceedings of the National Academy of Sciences (PNAS), discovered that the betrayal of this gene can occur in more ways than previously appreciated and ‘re-educating’ cells in the tumour microenvironment can reverse some of the malignancy.
All cells carry this gene, known as p53. This gene normally plays a central role in protecting the body against malignancy, orchestrating the cell’s defences against cancer and often killing a potentially cancerous cell if these fail.
In about half of cancer patients, the p53 gene within the cancerous cells contains alterations – mutations – that can result in the production of a p53 protein that not only fails to suppress cancer, but can even launch cancer-promoting activities.
But besides the cancerous cells, a malignant tumour contains a variety of non-cancerous cells and connective tissue elements, commonly referred to as the tumour microenvironment. In the initial stages of cancer development, the microenvironment is hostile to the tumour.
Professor Moshe Oren of Weizmann’s Molecular Cell Biology Department and other scientists found in earlier studies that the p53 of the microenvironment cells contributes to this hostility, blocking the spread of the cancer.
“This protective campaign probably often succeeds, otherwise people would get cancer much more frequently than they actually do,” said Professor Oren.
As the cancer progresses and becomes more malignant, the tumour microenvironment gradually changes. Scientists refer to this process as ‘education’: The microenvironment is being co-opted by the progressing tumour into promoting, rather than restricting, the cancer.
Among the co-opted cells are the fibroblasts, which supply the tissue with structural ‘cement’. Initially these help recruit immune cells against the cancer, but they now start releasing substances that encourage tumour growth, invasion and survival. At this stage, these cells are referred to as cancer-associated fibroblasts.
The new study, conducted in Oren’s lab in collaboration with Weizmann Institute colleagues, shows that the microenvironment’s ‘education’ – a more appropriate term would probably be ‘brainwashing’ – is directed in part at the fibroblasts’ p53.
As the cancer grows, the p53 in the fibroblasts switches sides. Although the p53 in the cancer-associated fibroblasts doesn’t acquire mutations as it does in the cancer cells, it nevertheless becomes altered in a manner that causes it to switch from restricting to supporting the cancer.
In the study, led by postdoctoral fellow Dr Sharath Chandra Arandkar, in collaboration with departmental colleague Professor Benjamin Geiger, and with Professor Yosef Yarden and Dr Igor Ulitsky of the Biological Regulation Department, the researchers showed that eliminating the p53 protein from cancer-associated fibroblasts by silencing their p53 genes caused these cells to lose many of their tumour-supporting features and behave more like normal fibroblasts.
In particular, the silencing of fibroblast p53 reduced the migration of adjacent cancer cells in a laboratory dish – a crucial change, considering that invasive migration facilitates the metastatic spread of cancer. Moreover, the silencing of p53 in cancer-associated fibroblasts greatly reduced the ability of these cells to promote tumour growth in mice.
Study authors included Weizmann’s Drs Noa Furth, Yair Elisha and Nishanth Belugali Nataraj, and, from Dr Margarete Fischer-Bosch Institute of Clinical Pharmacology in Stuttgart, Germany: Professor Walter Aulitzky and the late Dr Heiko van der Kuip, to whose memory this publication was dedicated.
Finding ways to ‘re-educate’ the renegade p53 in the tumour microenvironment – to reverse its behaviour back to suppressing tumours – may pave the way to the development of novel therapies that will target the microenvironment rather than the cancer cells themselves. Indeed, strategies targeting the cancer microenvironment are being increasingly explored in recent years. The hope is that they might provide a new window of opportunity for launching effective therapy, because the microenvironment tends to evolve more slowly than the mutation-ridden tumour cells.
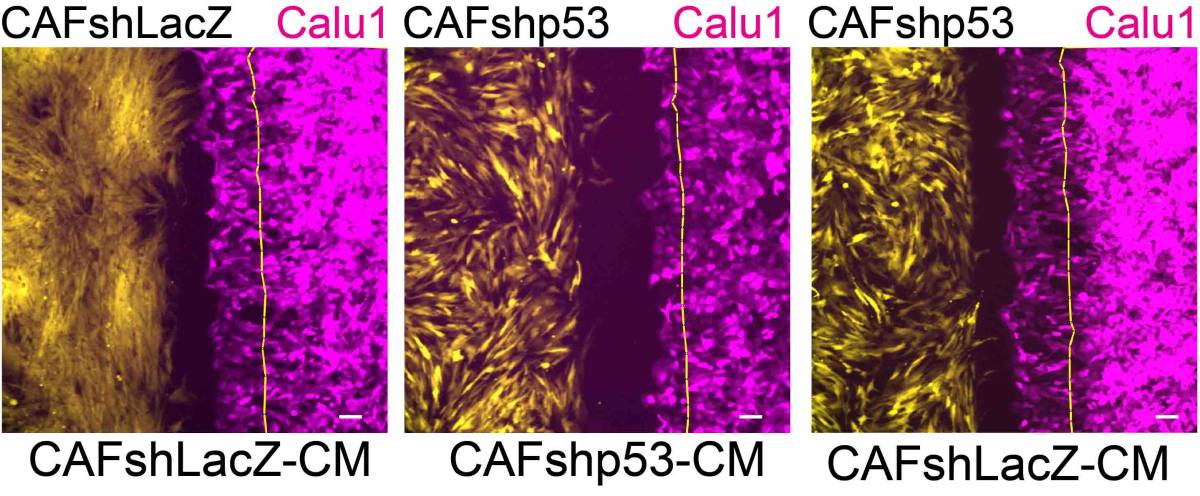
Cancer and it’s microenvironment: Time lapse movie of GFP-expressing immortalized NFshLacZ cells from patient 4731 (Yellow) and mCherry-expressing Calu1 cells (magenta). Cells were seeded in 12-well plates containing ibidi culture inserts. The next day, inserts were removed and cells were allowed to migrate. Images were captured every 15 min, for a total of 6 hrs.
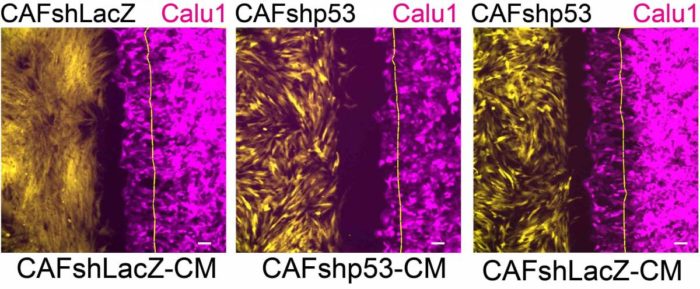
The effects of p53 in cancer-associated fibroblasts on cancer cell migration: Cancer cells (magenta) migrate in the direction of cancer-associated fibroblasts (yellow) that express a non-mutated p53 gene (left); this migration slows down (center) when the p53 in the fibroblasts is silenced; when substances released by the cancer-associated fibroblasts are added to the laboratory dish, the migration is restored (right)






