August 3, 2020
A new study in mice at the Weizmann Institute of Science show that the mother’s natural anti-viral proteins disrupted the development of neural circuits in the foetus.
Findings emerging from the study – recently published in Nature – showed that when a pregnant woman is infected with a virus, her immune system’s response to the infection could harm her baby’s brain – even if the foetus is not infected with the virus itself.
It’s been long known that a viral infection during pregnancy may increase the risk of schizophrenia, autism and other neurodevelopmental and neuropsychiatric disorders in her offspring later in life, but the exact mechanism of this effect is unknown. In particular, it’s been unclear whether the baby’s brain is liable to be harmed by the virus itself or by the pregnant woman’s immune response to the virus.
Professor Michal Schwartz of Weizmann’s Neurobiology Department and her team, in collaboration with the team of Professor Ido Amit of the Immunology Department, explored this question in this study.
The researchers had previously seen in their works that within the brain, large amounts of the interferon-beta protein – which, among its other functions, serves as the first line of defence against viral infection in mammals – can harm brain cells called microglia. These cells play an important role in embryonic development, helping to shape neuronal circuits; thus, the scientists designed a study focusing on microglia in the foetus.
They infected pregnant mice with a synthetic RNA molecule that mimics an infection caused by RNA viruses. The latter belong to a large family of viruses that induce diseases such as flu, measles, Ebola and COVID-19. When the mouse pups were born, it turned out that their brain’s immune cells, the microglia, were adversely affected and showed abnormal behaviour.
The researchers then set out to check whether this adverse effect on the microglia was caused by the virus-mimicking molecules or by the mother’s immune response to the viral infection – that is, by the antiviral immune system’s protein, interferon-beta, whose levels rise sharply as a result of this infection. They exposed the pregnant mice to the virus-mimicking molecules but at the same time treated them with antibodies that neutralize the interferon-beta. The treatment reduced the adverse effect on the microglia of the pups, suggesting that this effect was due to the interferon-beta.
Next, to double check this conclusion, the scientists – instead of infecting the pregnant mice with the virus-mimicking molecules – injected them with the interferon-beta. In this case, too, the microglia of the newborn pups displayed similar abnormal functioning. Moreover, the pups themselves were later shown to behave abnormally, in a manner similar to the one exhibited in neuropsychiatric disorders, and they were more prone to stress than those born to mothers that had not been exposed to high levels of interferon-beta.
These results show that even when the foetus is not directly exposed to a virus found in the mother’s body, the microglia in the fetal brain might be damaged by the interferon-beta that the mother secretes in large amounts in response to the viral infection.
“Further research may find ways of protecting the foetus from the mother’s response to the virus – that is, from the interferon that rises during viral infection,” said Schwartz.
“In the meantime, pregnant women would do well to exercise caution, so as to avoid becoming infected with viruses during pregnancy.”
The study was carried out by Dr Hila Ben-Yehuda with the assistance of several former members of the two teams, including Dr Alexander Kertser of the Neurobiology Department, and Dr Orit Matcovitch-Natan and Amit Spinrad of the Neurobiology and Immunology Departments.
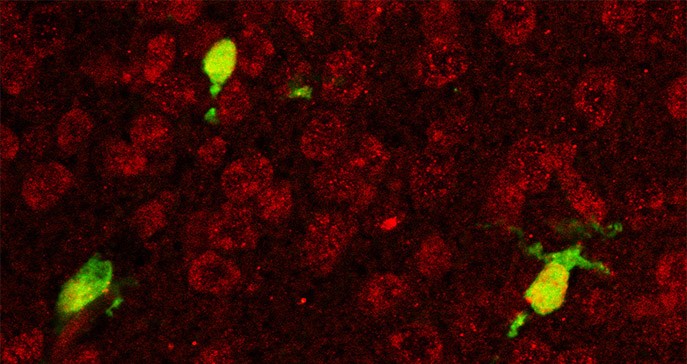
A combined microscope image reveals that the microglia (green) in a newborn mouse brain express interferon receptors (red)

Professors Michal Schwartz and Ido Amit