January 8, 2020
A host of disease-causing viruses called ‘arenaviruses’ lurk in animal populations in various parts of the world, sometimes crossing over into humans. When they do cross over, they can be lethal, and few treatments exist.
Now researchers at the Weizmann Institute of Science have devised a clever decoy for these viruses that may keep them from spreading in the body.
Two disease-causing arenaviruses, known as Junín and Machupo, circulate through rodent populations, mainly in South America, and they can infect humans when people come in contact with affected rodents. Similar to Ebola, these diseases can cause the body to ‘bleed out’, and the only treatments, to date, are risky and complex, as they are taken from the blood of survivors.
The study, reported in Nature Communications, arose from an earlier research question in the group of Dr Ron Diskin of the Weizmann Institute’s Structural Biology Department: how are certain arenaviruses able to move from rodents or other animals into humans?
Comparing these viruses with members of the arenavirus family, not infectious to humans, the researchers noted that the non-infectious viruses did not completely fit a particular receptor – a protein complex on the cell membrane – that serves as a viral entry point into human cells. But curiously, those that do infect humans were also not a perfect fit. They were just good enough to get by.
This observation led to another insight: Maybe the rodent cell receptors, which were a much better fit to the ‘entry’ proteins on the viruses, could be used to intercept the viruses and lure them away from the human cells.
Diskin explained that there had been earlier attempts to develop such decoys – ‘sticky’ molecules designed to attract virus proteins, but these were based on the structures of human receptors, so they were identical to the ones on the body’s cells and thus unable to compete effectively. In contrast, a molecule based on a rodent receptor, he and his team reasoned, could far outcompete the human ones for binding to the virus.
Dr Hadas Cohen-Dvashi, in Diskin’s group, led the next stage of the research, in which she ‘surgically removed’ the very tip of the rodent receptor to which the virus binds and engineered it onto part of an antibody. The newly resulting molecule was called ‘Arenacept’.
Then the group began testing this molecule – at first against ‘pseudoviruses’, engineered virus-like complexes which carry the entry proteins but that are not dangerous. Already at this stage, and in collaboration with the group of Dr Vered Paler-Karavani of Tel Aviv University, the researchers noted that Arenacept not only bound strongly to the viruses, it recruited parts of the immune system to mount an attack against the viral invasion.
The next stages of testing took place in labs in the University of Texas, Galveston, in the United States, and in the Pasteur Institute, Lyon, France, which are equipped to test pathogens at the highest safety levels (BSL-4). Here, Arenacept was pitted against human receptors in lab tests simulating attacks by two of the real pathogenic viruses – the Junín and Machupo viruses.
The researchers found Arenacept to be highly effective at sticking strongly to the viruses before they could bind to the human receptors, and as with the pseudoviruses, they noted the activation of the immune response.
Because Arenacept is based on the entry point shared by all viruses in a given family, rather than on individual characteristics of each virus (as is the case with vaccines or antibodies), it should be equally effective against all the viruses in this family that cross to humans from animals and for that utilize the same receptor.
“It might even be effective against viruses from the same family that have not yet been discovered or newly-emergent ones,” said Diskin.
“All signs suggest Arenacept is non-toxic, and that it is also heat-resistant and stable, meaning it could be delivered to the remote areas where these diseases are endemic,” he said.
“Also the idea of creating decoys from mammal receptors might be applied to all sorts of other diseases that cross to humans from animals.”
Yeda Research and Development, the technology transfer arm of the Weizmann Institute of Science, has received a patent for Arenacept and is working with Diskin to advance clinical research.
Also participating in this research were Aliza Borenstein-Katz of the Weizmann Institute of Science; Ron Amon of Tel Aviv University; Krystle N. Agans, Robert W. Cross and Thomas W. Geisbert of the University of Texas; and Mathieu Mateo, Sylvain Baize of the Pasteur Institute.
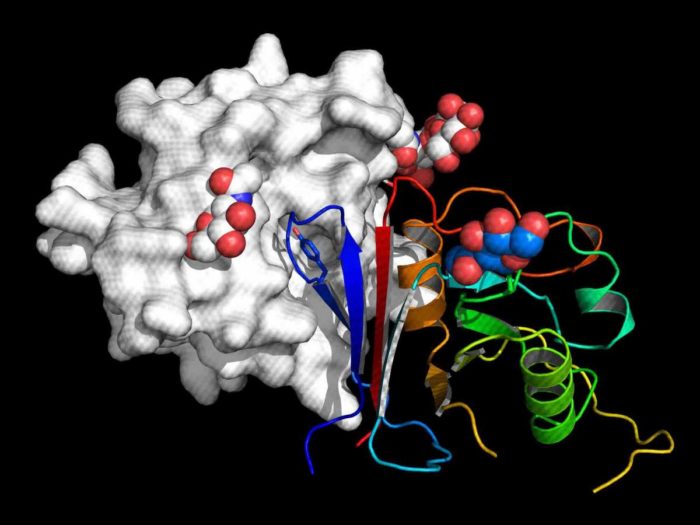
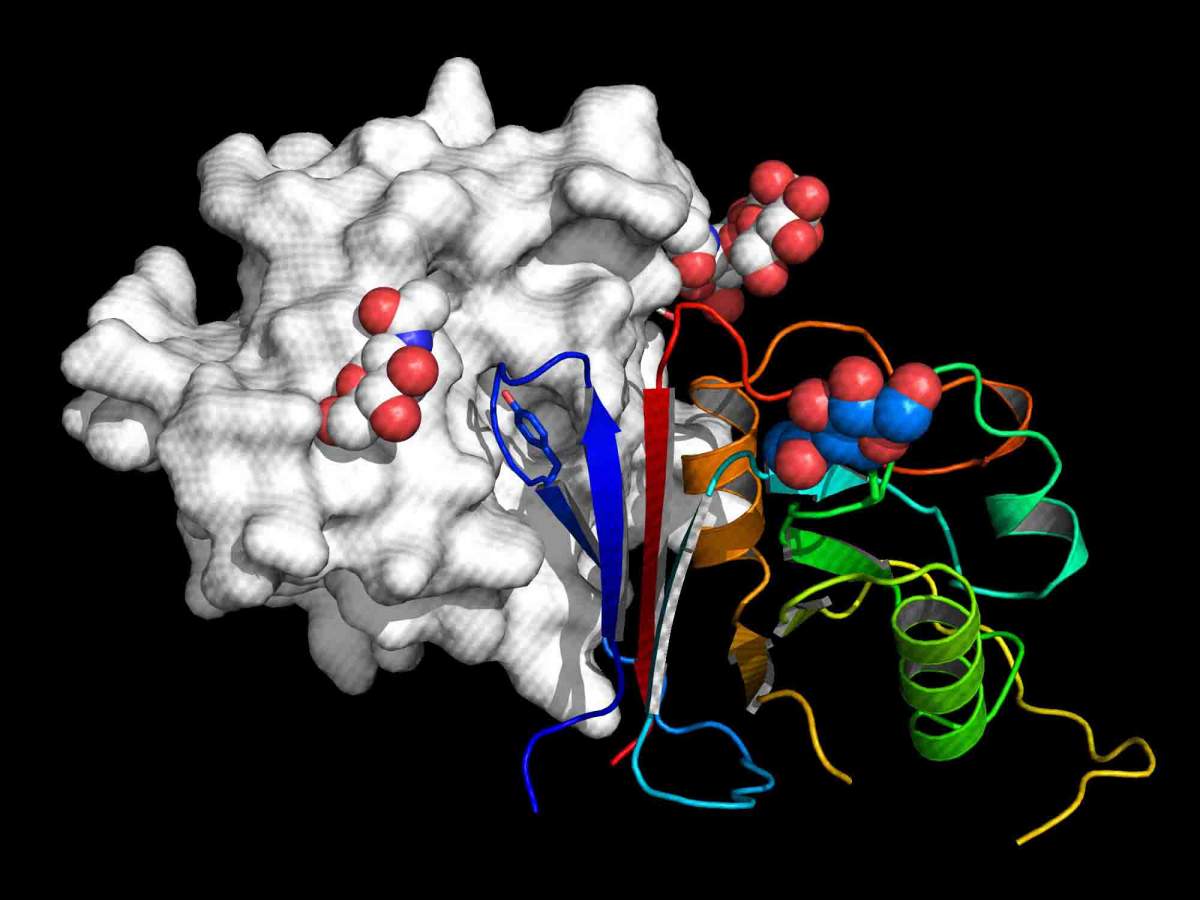
The active part of Arenacept is shown as rainbow-colored ribbon bound to the receptor binding domain of Macho virus (grey surface representation)