February 19, 2017
If you get up in the morning feeling energetic and clearheaded, you can thank your liver for manufacturing glucose before breakfast time. Among a host of other vital functions, it also clears our body of toxins and produces most of the carrier proteins in our blood.
In a study reported recently in Nature, Weizmann Institute of Science researchers showed that the liver’s amazing multitasking capacity is due at least in part to a clever division of labor among its cells.
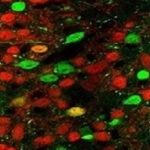
Each of the liver’s microscopic, hexagonal lobules consists of onion-like concentric layers. By mapping gene activity in all the cells of a liver lobule, Dr Shalev Itzkovitz of Weizmann’s Molecular Cell Biology Department and his research team have revealed that these layers each perform different functions.
“We’ve found that liver cells can be divided into at least nine different types, each specializing in its own tasks,” said Itzkovitz.
The scientists found, for instance, that the synthesis of glucose, blood-clotting factors and various other materials takes place in the outer layers of the liver lobule.
“These layers are rich in the oxygen needed to fuel these costly synthesis processes,” explained Itzkovitz.
The inner layers of the liver lobules revealed themselves to be the sites where toxins and other substances are broken down.
The middle layers also proved to have their own functions, rather than serving as mere transition zones. The researchers found, for example, that cells in these layers manufacture the hormone hepcidin, which regulates iron levels in the blood.
The scientists also discovered that certain processes, such as the manufacture of bile, proceed across several different layers, in something like a production line.
These discoveries emerged when the researchers created a spatial atlas of gene expression for all liver cells, the first of its kind for this organ.
In collaboration with Professor Ido Amit of Weizmann’s Immunology Department, they analyzed the genomes of 1,500 individual liver cells, establishing patterns of expression for about 20,000 genes in each cell.
In parallel they visualised intact liver tissue, locating individual messenger RNA molecules under a fluorescence microscope, using a method developed by Itzkovitz and his colleagues. Special algorithms then enabled the researchers to establish both the gene expression in each cell and the location of these cells in the liver lobule. They found that more than half of the 7,000 genes expressed in the liver vary in activity from one layer to another, a number that is about ten times greater than previous estimates.
Such an in-depth analysis of gene expression may help clarify the course and origin of common liver disorders, including liver cancer and non-alcoholic fatty liver disease, which affects about a fifth of the population in developed countries. In addition the approach developed in the new study may now be applied to map gene expression elsewhere in the body.
The research team included postdoctoral fellows Drs Keren Bahar Halpern, Beata Toth, Doron Lemze and Andreas E. Moor and graduate students Rom Shenhav, Matan Golan, Efi Massasa, Shaked Baydatch, Shanie Landen and Avigail Stokar-Avihail of the Molecular Cell Biology Department, as well as graduate students Orit Matcovitch-Natan, Amir Giladi and Eyal David from Professor Amit’s lab in the Immunology Department and staff scientist Dr. Alexander Brandis of Weizmann’s Life Sciences Core Facilities.
Dr Shalev Itzkovitz’s research is supported by the Henry Chanoch Krenter Institute for Biomedical Imaging and Genomics; the Rothschild Caesarea Foundation; the Cymerman – Jakubskind Prize; and the European Research Council. Dr Itzkovitz is the incumbent of the Philip Harris and Gerald Ronson Career Development Chair.
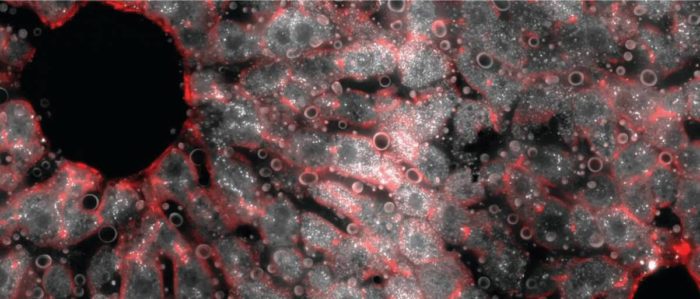
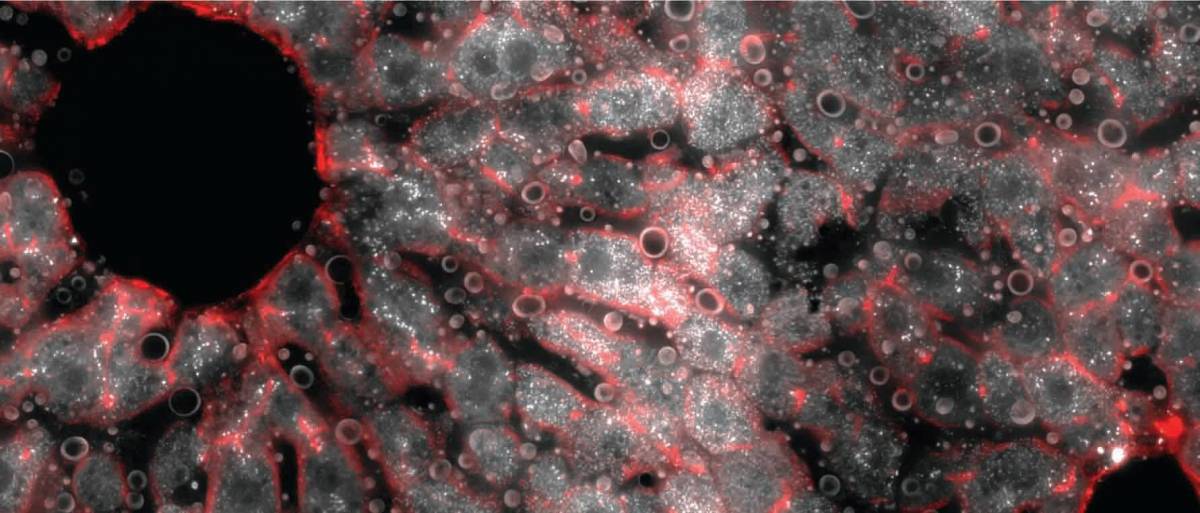
A cross section of a mouse liver lobule under a fluorescence microscope. The middle layer reveals an abundance of messenger RNA molecules (white dots) for the gene encoding hepcidin, the iron-regulating hormone.