
June 5, 2024
Researchers from the University of Michigan and the Weizmann Institute of Science have developed the first-ever complete central nervous system on a chip, emulating that of a human embryo, from the forebrain to the bottom of the spinal cord.
Once upon a time, we were all nothing but a mass of densely packed stem cells. Over time, this mass elongated, sprouted limbs on either side, buttocks at the rear, a stomach in the front and a head on the top.
The process by which embryonic stem cells give rise to distinct organs, rescuing us from an amorphous fate, occurs thanks to morphogens, molecules that are manufactured at specific times and places within the embryo and dispersed to dictate the location and shape of our organs. The varying concentrations of morphogens serve as a map that guides the stem cells to their destination and destiny.
Morphogen concentration maps are key to all technologies aimed at making organoids, the laboratory-manufactured miniature versions of living organs that in the past decade have taken over the world of developmental biology. But until now, most researchers have been producing organoids using uniform concentrations of morphogens in petri dishes, which limited them to growing small sections of an organ in each dish, rather than generating a single, miniature version of the complete organ.
Now, researchers from the University of Michigan, who were led by Professor Jianping Fu and Dr Xufeng Xue, from the Weizmann Institute of Science and from the University of Pennsylvania have created a miniature version of the entire embryonic central nervous system, from the brain to the bottom of the spinal cord, using a microfluidic chip that mimics the dispersal of the morphogens during embryonic development.
The new chip will enable researchers to ask entirely new questions, both about the development of a healthy embryo and about diseases and tissue damage, according to study participant Professor Orly Reiner of Weizmann’s Molecular Genetics Department. She has been studying diseases affecting the developing brain for more than 30 years and started growing organoids in her laboratory about a decade ago.
“Organoids were already exposed to varying concentrations of morphogens in past studies, but those studies generated only small sections of the central nervous system – for example, just the spinal cord or the forebrain, but not both,” she explained.
The microfluidic chip allows researchers to pour morphogens, from almost any direction and at any time they want, into reservoirs that contain the organoids. At the centre of the chip are narrow, adhesive surfaces that are 4 millimetres long, just like the central nervous system of a month-old embryo. Stem cells are embedded along the length of these surfaces, which are then covered with a gel that simulates the extracellular environment, allowing them to develop into three-dimensional tissue.
Within a short period of time, the cells spontaneously organise themselves into a hollow tube. After three days, through a reservoir on one end of the chip, the researchers start adding morphogens, which diffuse slowly across the length or breadth of the tissue.
The researchers soon saw that the stem cells on the chip matured into a variety of different cell types of the embryonic central nervous system. The side of the chip with the high concentration of morphogens gave rise to cells that developed into the end of the spinal cord, followed by the cells destined for the middle of the spinal cord, the hindbrain, the midbrain and, at the furthest extremity, the forebrain.
“When we characterised the new organoids, we saw perfect order along the entire length of the central nervous system, just as it appears in the early embryonic stage,” Reiner said.
Having created the central nervous system’s longitudinal tube, the researchers took on another challenge: to emulate the development of the embryo’s forebrain along the ventral-dorsal axis. Two important cell types that are essential to the functioning of the adult brain are usually generated in the forebrain: excitatory neurons, which encourage neuronal firing, and inhibitory neurons, which block this firing.
“Until now, we had to grow each of these cell types on a different plate by exposing two organoids to different morphogen concentrations, and then try to join them together,” Reiner explained.
But the new microfluidic chip enabled the researchers to distribute the concentrations of morphogens so that the two types of neurons were generated in the same tissue.
To do this, they first created the longitudinal tube, as described above, and on the seventh day they poured in morphogens near the forebrain but far from the spinal cord.
Within a short period of time, cells that would later turn into inhibitors appeared on the inside of the tube, while those destined to become excitatory cells appeared on the outside of the tube – exactly as happens during embryonic development.
The researchers note that their chip does not emulate the earliest stages of the central nervous system’s development.
“We are actually skipping over the early stages and pushing the stem cells to the stage of development typical of a four-week-old embryo,” Reiner said.
Still, within days a three-dimensional tissue formed, it bore a remarkable resemblance to the embryo’s central nervous system, both in terms of the cells it contained and the order of their appearance. This allowed the team to study, for example, the genes involved in the differentiation of cell populations in the spinal cord, a process that was not previously understood.
The chip is already helping researchers further their understanding of issues related to the development of the human nervous system. Reiner’s team, for one, has integrated the technology into its work and is using the chip to study how genetic diseases affect the longitudinal development of parts of the brain. They hope that more researchers will use the technology to expand our understanding of a wide range of diseases that damage the nervous system.
Also participating in the study were Dr Yung Su Kim, Dr Norio Kobayashi, Dr Yue Liu, Dr Jason R. Spence, Dr Robin Zhexuan Yan, Dr Yu-Hwai Tsai, Shiyu Sun and Yi Zheng from the University of Michigan; Dr Rami Yair Tshuva and Alfredo-Isaac Ponce-Arias from Weizmann’s Molecular Genetics Department; Professor Hongjun Song, Professor Guo-Li Ming and Dr Richard O’Laughlin from the University of Pennsylvania; and Professor Azim Surani and Dr Frederick C. K. Wong from Cambridge University.

(l-r) Alfredo-Isaac Ponce-Arias and Professor Orly Reiner of Weizmann’s Molecular Genetics Department
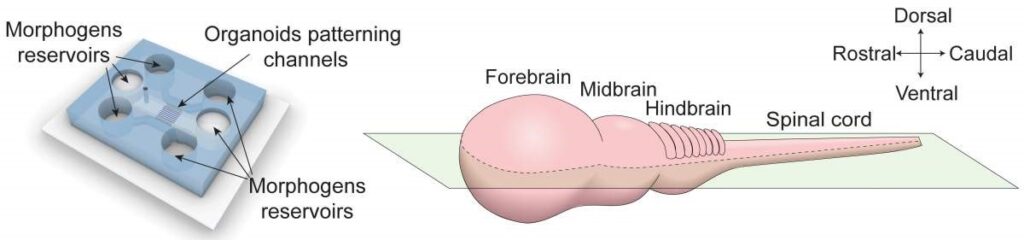
Left: A diagram of the new microfluidic chip that allows researchers to create varying morphogen concentrations. Right: A diagram of the embryo’s central nervous system
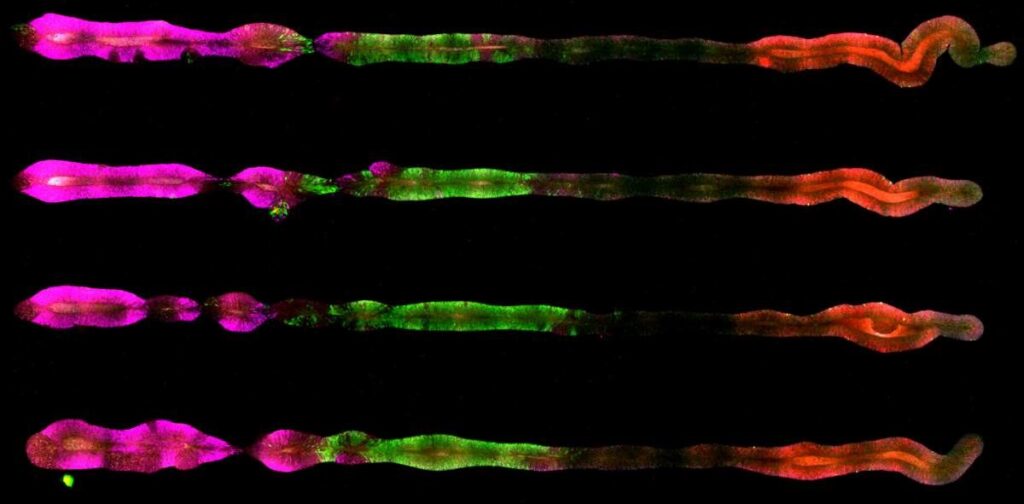
Researchers used color-coded proteins to reveal the identity of the cells in the organoids they produced. In four organoids that simulate the embryo’s central nervous system, the magenta marks proteins associated with the development of the forebrain and the midbrain, the green, the hindbrain and the red, the center of the spinal cord

Professor Jianping Fu of the University of Michigan's Mechanical Engineering Department in his lab with a research student. Photo by Marcin Szczepanski, Michigan Engineering






