
August 1, 2024
New research from the Weizmann Institute of Science – just published in Nature – shows that antibody-producing cells discovered inside the nasal conchae may help to develop new nasal vaccinations and treatments for allergies and nervous system disorders.
The nose is a major gateway to our bodies – for the air we breathe, the aromas we smell and the microbes that make us sick. On its way in, the air passes through nasal conchae, or turbinates – the long, narrow, curled shelves of bone that look like a shell and protrude into the breathing passage. The conchae are covered by a unique kind of tissue that secretes mucus and contains many branches of nerve cells that are responsible for our sense of smell. The structure and function of the conchae allows the air to warm up and absorb moisture before reaching the lungs.
Not only do the nasal conchae serve as a leading site for pathogen invasion into the airways, but they also have a major weak spot: Because they are located so close to the brain, they are not accessible to the antibodies dispatched by our immune system via the blood stream during an upper airway infection. How, then, are we relatively protected from invading microbes and not constantly sick?
In the new study, the Weizmann researchers found that antibody-secreting cells migrate to the nasal conchae whenever we are sick or given a vaccination, and from there they secrete antibodies locally into the nasal cavity. This discovery could pave the way for more effective nasal vaccinations and new treatments for nervous system disorders, allergies and autoimmune diseases.
During the coronavirus pandemic, when the virus was spreading across the globe, millions of people kept close tabs on the development of a COVID vaccine that could be administered as a nasal spray instead of an injection. The idea was not outlandish; after all, a flu vaccine and several others are already available through nasal sprays, which contain live but weakened viruses that generate local protection in the nasal conchae. These nasal vaccinations, however, are not effective in a single dose and must be followed up by a booster dose. Yet, scientists do not yet fully understand how these nasal vaccines work and why a booster is required.
To shed light on the matter, Professor Ziv Shulman of Weizmann’s Systems Immunology Department and Jingjing Liu, a PhD student in his lab, decided to examine how immune system organs located near the nose and throat respond to nasal vaccinations. In humans these organs include the tonsils and the adenoids, collectively known as Waldeyer’s lymphatic ring, or tonsillar ring. In this new study, a team of researchers, led by Liu, used advanced imaging techniques to analyse the body’s immune response by imaging whole and intact immune system organs of mice, similar to those of humans.
The researchers discovered that when mice were given a nasal vaccination, a focused immune response was mounted by B cells, the immune system’s key producers of antibodies. These cells begin their journey as precursor B cells, some of which have the potential to identify pathogens.
The scientists saw how cells located close to the mucous tissue that covers the nasal cavity identified the vaccine molecules and started to divide and differentiate rapidly. This differentiation process can be seen as a kind of biological specialization course, which ends when they become cells that secrete antibodies specific to the pathogen or become memory cells. The memory cells are stored for extended periods of time in case of future infection.
B cells can secrete five types of antibodies. The researchers discovered that in response to a vaccine the B cells close to the nasal cavity change the identity of the antibody that they produce and start secreting antibodies that act as ‘gatekeepers’ that specialize in passing from the inner mucous tissue into the nasal cavity. The next stage is for the B cells to move from their location close to the nasal cavity to tiny ‘training camps’ known as germinal centres, which are found in the immune system organs in that part of the body. Once there, they undergo “training” that includes changes to their genetic make-up and a meticulous selection process to ensure the survival of only those B cells that produce the antibodies effectively linked to the pathogen targeted by the vaccine.
The B cells are aided in this training plan by a certain type of T cell, which even plays a role in deciding which cells will survive in the end, but the researchers discovered that there are not enough of these T cells in the organs of the immune system in that part of the body to create an effective immune response.
“The fact that there has to be a migration of T cells to the area explains why one dose of a nasal vaccine is not enough and a booster dose is required,” Shulman explained.
“It is only after the second dose that enough of the required T cells are produced in order to turn the B cells into effective antibody producers and into memory cells.”
The researchers believe that the scarcity of T cells is designed to prevent oversensitivity to harmless quantities of foreign objects in the air.
“It is possible that this mechanism goes awry when people develop allergies and various autoimmune diseases, so understanding it could help develop new treatments for these conditions,” Shulman added.
An immunisation treasure hunt
Discovering that the immune mechanism was activated in response to vaccines was only the starting point for an intensive search to determine where the antibody-secreting cells go after the differentiation process is completed.
“We visualised an immune response in the nasal lymph nodes, but how is it translated into airway protection?” said Shulman.
“We were surprised to discover the B cells in the nasal conchae, bone tissue that was not known to support an antibody-mediated immune response. This relocation to bone tissue is similar to what happens in bone marrow, and it is possible that this niche environment has other roles beyond the immune mechanism that we identified.”
The new findings show that the antibody-secreting cells translocate themselves from the nasal lymph nodes to the mucus-producing glands in the nasal conchae, directly below their external layers of cells, and that they secrete their antibodies into these glands. This immune defence makes up for the blood antibodies’ inability to get to this gateway through the blood stream, and it is important not only in the context of viruses and other diseases: It also protects the brain and the many nerve endings in that part of the body, which are responsible for our sense of smell.
In addition to facilitating vaccine design, Shulman noted that this research has exposed “an entry point into a highly fortified target – antibody-secreting cells that have access to the central nervous system”. In the future it might be possible to make use of the antibody-secreting cells’ access to the olfactory nerves in order to design vaccines for neurological diseases.
Also participating in the study were Dr Liat Stoler-Barak, Dr Hadas Hezroni-Bravyi and Dr Adi Biram from Weizmann’s Systems Immunology Department; Sacha Lebon, Dr Natalia Davidzohn and Dr Moshe Biton from Weizmann’s Immunology and Regenerative Biology Department; Dr Merav Kedmi, Muriel Chemla and Dr David Pilzer from Weizmann’s Life Sciences Core Facilities Department and the Nancy and Stephen Grand Israel National Center for Personalized Medicine at the Weizmann Institute; and Dr Marina Cohen and Dr Ori Brenner from Weizmann’s Veterinary Resources Department.

Professor Ziv Shulman
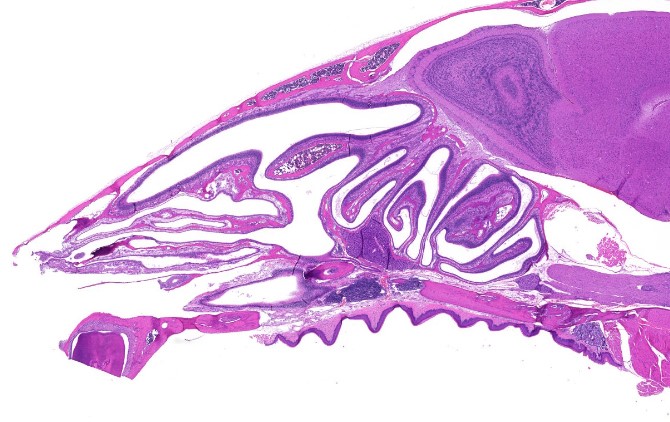
Cross-section of the nasal cavity and conchae of a mouse that was given a nasal vaccination

(l-r) Jingjing Liu and Dr Liat Stoler-Barak

B cells (gold) inside the nasal conchae (green) of a mouse that has received a nasal vaccination






