New research from the Weizmann Institute of Science published in Cell Reports and recently reported in The Times of Israel, indicates that oxygen starvation could toughen up immune T cells for cancer immunotherapy.
So it is not just mountain climbers and endurance athletes that can benefit from ‘altitude training’ by learning to perform well under low-oxygen conditions. These cancer-fighting cells of the immune system can also improve their performance through a cellular version of such a regimen.
Harnessing the immune system to battle malignancy – an approach known as cancer immunotherapy – has already started saving the lives of cancer patients in the past few years. In one major version of this approach, killer T cells are removed from the patient’s blood, grown in a laboratory dish and adapted to identifying and destroying cancerous cells; they are then returned to the patient’s bloodstream. This method has so far worked best against certain leukemias and lymphomas, but not against solid tumours, possibly because within such tumours, oxygen concentration is extremely low: 0.5% to 5% of the gas dissolved in the extracellular fluid – lower than in most healthy organs, and certainly much lower compared with a regular lab incubator, in which oxygen accounts for 20% of the gas dissolved in the culture fluid used for growing the cells.
Tumour cells don’t seem to mind the shortage; they manage to make effective use of glucose, the major cellular fuel, even when oxygen concentration is low. But the same is not true for T cells, which have a hard time penetrating tumors and performing their killing function. Previous studies had shown that growing T cells under low-oxygen conditions helps them kill other cells in a laboratory dish, but their actual cancer-fighting ability has never been tested.
“Killer T cells are the foot soldiers of cancer immunotherapy, they are the ones to target and destroy cancerous cells, but they don’t always manage to eliminate the malignancy,” said team leader Professor Guy Shakhar of Weizmann’s Immunology Department.
“We’ve shown that by growing these T cells in an oxygen-poor environment, we can turn them into more effective killers.”
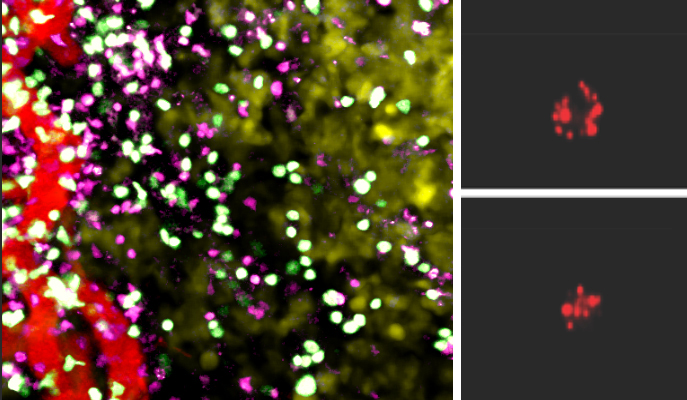
In the new study, research student Yael Gropper and staff scientist Revital Zehavi-Feferman from Shakhar’s team, together with Drs Tomer Meir Salame and Ziv Porat of Weizmann’s Life Sciences Core Facilities Department, and Dr Tali Shalit from the Grand Israel National Center for Personalized Medicine, put T cells through an altitude training of sorts – by growing them in an incubator with an oxygen concentration as low as 1%. They then divided mice affected by melanoma tumors into two groups; one group was injected with the oxygen-starved T cells, the other with T cells grown under regular oxygen conditions.
Oxygen-starved T cells proved much more effective at fighting the cancer. Mice treated with these cells lived longer and their tumors shrank much more dramatically compared with the mice treated with regular T cells. Surprisingly, the oxygen-starved T cells did not penetrate the tumors better than did the regular cells. Apparently, they countered the cancer more successfully because they had a higher content of the destructive enzyme, known as granzyme B, that penetrates and kills cancerous cells.
“Just as altitude training increases endurance in humans, so putting killer T cells through a ‘fitness regimen’ apparently toughens them up,” Shakhar said.
If these findings are confirmed with human T cells, they may provide an immediate means of improving immunotherapy against solid tumours.
“In cellular immunotherapy, T cells need to be removed and grown outside the body in any event. Growing them under low oxygen pressure is relatively simple, but this small adjustment to existing clinical protocols may significantly improve the therapy’s effectiveness,” concluded Shakhar.
###